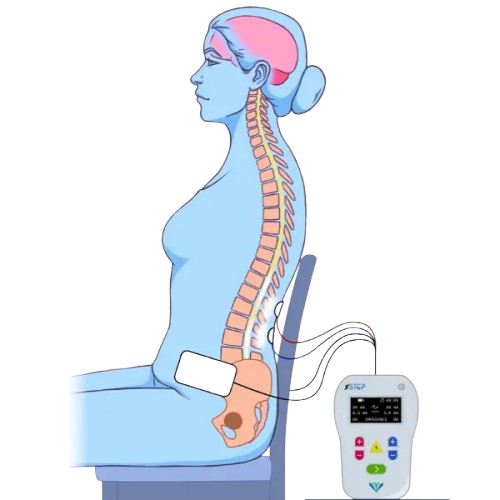
xStep is a neurotechnology platform using non-invasive spinal neuromodulation to restore movement, sensation, and functional independence in individuals with neurological conditions.
Built on clinically validated SCiP and SCONE technologies and developed by VIVATRONIX, xStep enables the nervous system to relearn and reorganize through targeted stimulation and therapy.
Designed for conditions such as spinal cord injury, stroke, and cerebral palsy, xStep activates dormant neural pathways and supports measurable improvements in motor function, sensation, and autonomic control.